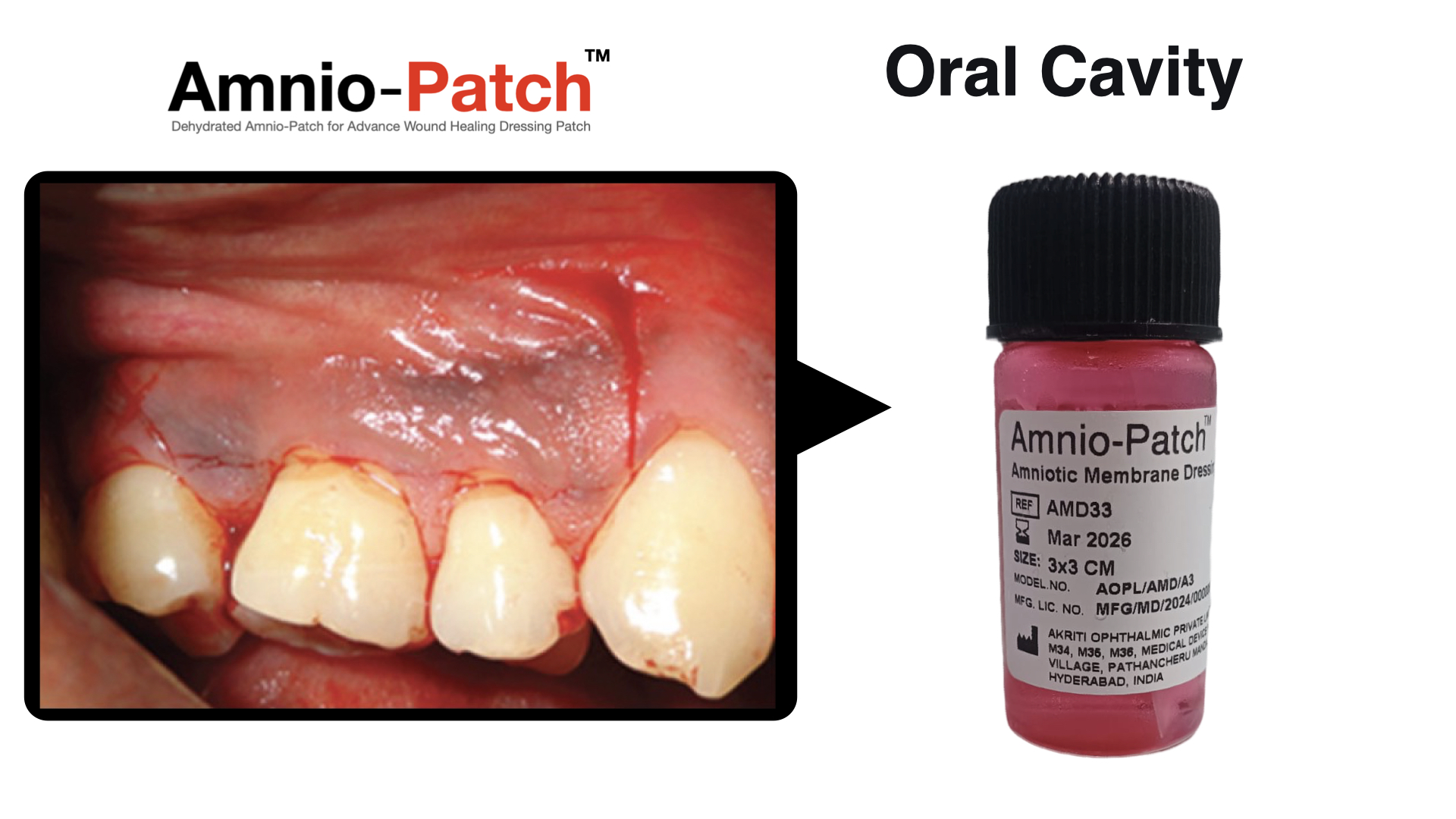
Orthopedics, Regenerative Medicine & Immunology
Amnio‑Patch‑C™ is a cryogenically preserved human amniotic membrane allograft, processed under stringent quality systems consistent with 21 CFR 1271.10(a) and regulated solely under Section 361 of the Public Health Service Act.
Cryopreservation preserves the native extracellular matrix, growth factors, and bioactive components, supporting anti‑inflammatory, anti‑fibrotic, and regenerative functions across multiple medical specialties.
Its biologically active scaffold makes it a valuable adjunct in orthopedic repair, tissue engineering, and immune‑modulating applications.
Orthopedics
1. Soft Tissue Repair
Amnio‑Patch‑C™ can be used as a biological covering or interpositional graft in soft‑tissue orthopedic procedures, including:
Tendon injuries (e.g., rotator cuff, Achilles tendon)
Muscle repairs following trauma or surgical release
Ligament reconstruction (e.g., ACL, collateral ligaments)
Clinical benefits:
Reduction of inflammation at the repair site
Support for organized collagen deposition
Decreased scar tissue formation
Enhanced gliding surface for tendons and ligaments
These properties help improve functional recovery and reduce post‑operative adhesions.
2. Cartilage Repair
Emerging studies support the use of amniotic membrane in cartilage defect repair, including:
Focal chondral defects
Early osteochondral degeneration
Adjunct to microfracture or scaffold‑based procedures
Rationale:
The membrane’s extracellular matrix supports cell adhesion, proliferation, and differentiation, making it a promising biologic scaffold for cartilage regeneration.
Regenerative Medicine
3. Tissue Engineering Applications
Amnio‑Patch‑C™ serves as a biocompatible scaffold in tissue engineering due to its structural and biochemical properties.
It supports:
Cell proliferation
Stem cell attachment and differentiation
Formation of organized new tissue
Reduced inflammatory response during regeneration
Applications include:
Musculoskeletal tissue engineering
Skin and soft‑tissue reconstruction
Bioengineered graft development
Its natural ECM composition makes it suitable for both in vivo and in vitro regenerative platforms.
Immunology
4. Immune Modulation
The bioactive components of Amnio‑Patch‑C™—including cytokines, growth factors, and anti‑inflammatory mediators—contribute to immune regulatory effects.
Potential clinical relevance includes:
Modulation of autoimmune‑driven inflammation
Supportive therapy in immune‑mediated soft‑tissue disorders
Adjunctive use in conditions involving graft‑versus‑host inflammatory responses
These effects arise from the membrane’s ability to:
Down‑regulate pro‑inflammatory cytokines
Promote a balanced healing environment